Complications related to hernia repair involving surgical mesh have been reported to the FDA. A large number of these claims were associated with mesh products that are no longer on the market.
One such mesh product is Physiomesh™, manufactured by Johnson & Johnson subsidiary Ethicon.
On May 27, 2016, Ethicon initiated a withdrawal of its Physiomesh™ Flexible Composite Mesh (for laparoscopic use) from the worldwide market. The company sent an official letter to doctors advising them about problems with the mesh and the market withdrawal. Unfortunately, Physiomesh™ patients were not on Ethicon’s mailing list. Ethicon decided to withdraw Physiomesh™ based on an analysis of unpublished data from two large independent hernia registries.
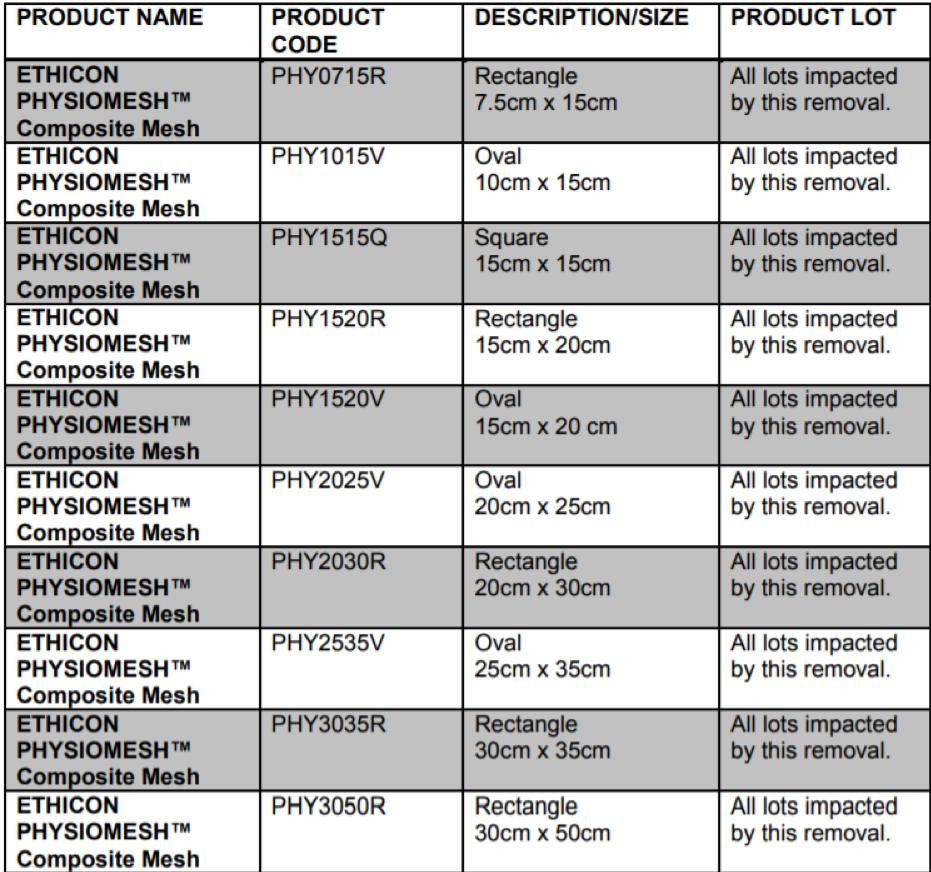
The recurrence/reoperation rates after laparoscopic ventral hernia repair using Ethicon Physiomesh™ Composite Mesh were higher than the average rates of the comparator set of meshes among patients in these registries. Ethicon, therefore, decided to remove its Physiomesh™ Composite Mesh from the global market. The products impacted by this market withdrawal are the following:
What Is Hernia Mesh?
Because hernias have a high recurrence rate, surgeons often use surgical mesh to strengthen the hernia repair and reduce the recurrence rate. There are two kinds of hernia mesh—absorbable and non-absorbable mesh.
Absorbable mesh is typically made of animal tissue processed for medical use. This type of mesh is designed to disintegrate as new tissue grows to strengthen the hernia repair.
Non-absorbable mesh is made of synthetics and is designed to provide long-term reinforcement to the hernia repair site.
Some types of hernia mesh combine absorbable with non-absorbable material, though these are rare. Hernia mesh products are designed to repair hernias and other defects by reinforcing the surrounding soft tissue.
What Is a Hernia?
A hernia occurs when an organ, intestine, or fatty tissue squeezes through a hole or a weak spot in the surrounding muscle or connective tissue. Hernias often occur at the abdominal wall. However, sometimes a hernia can be visible as an external bulge, particularly when straining or bearing down.
The Most Common Types of Hernias Include:
- Inguinal: occurs in the inner groin
- Femoral: occurs in the upper thigh/outer groin
- Incisional: occurs through an incision or scar in the abdomen
- Ventral: occurs in the general abdominal/ventral wall
- Umbilical: occurs at the belly button
- Hiatal: occurs inside the abdomen, along the upper stomach/diaphragm
What Causes Hernias?
A combination of pressure and an opening or weakness of muscle or connective tissue causes most hernias. The pressure pushes an organ or tissue through the opening or weak spot. Sometimes, muscle weakness is present at birth, but more often, it occurs later in life.
Anything that causes an increase in abdominal pressure can cause a hernia, including obesity, lifting heavy objects, diarrhea or constipation, or persistent coughing or sneezing. In addition, poor nutrition, smoking, and overexertion can weaken muscles and contribute to the likelihood of a hernia.
Surgical Treatment Options for Hernias
Laparoscopic: The surgeon makes several small incisions in the abdomen that allow surgical tools into the openings to repair the hernia. Clinicians may perform laparoscopic surgery with or without surgical mesh.
Open Repair: The surgeon makes an incision near the hernia to repair the weak muscle area. The clinician can perform an open repair with or without surgical mesh. Open repair that uses sutures without mesh is referred to as primary closure. Primary closure is used to repair inguinal hernias in infants, small hernias, strangulated or infected hernias.
What Are The Symptoms of a Failed Hernia Mesh Implant?
All patients who were implanted with Physiomesh™ are at an increased risk of serious complications, making it critical that you and your healthcare team monitor symptoms of failed mesh.
These symptoms include:
- Pain
- Swelling
- Heat or soreness around the surgical site
- Difficulties passing stool
- Fever and nausea
- Infection
- Rejection/Failure to incorporate
- Wound dehiscence
- Chronic seroma
- Chronic and severe pain
- Bowel adhesions/blockage
- Sinus tract or fistula formation
- Organ perforation
Do I Qualify to File an Ethicon Hernia Mesh Lawsuit?
If you received an Ethicon Physiomesh™ hernia mesh implant and are experiencing any of the symptoms we mentioned, you may be eligible for compensation.
Our law firm has a long history of helping patients injured or harmed by dangerous medical devices, such as Physiomesh™ and can help you join a Ethicon Mesh Class Action lawsuit to help cover damages.
Hiring an attorney from Meshbesher & Spence means you will benefit from our experience and dedication. It also means if you do not recover compensation for your injuries, you pay nothing.